Monday’s Lesson: Inquiry in the Digital Classroom
I hate the phrase ‘inquiry-based science’… It’s like saying ‘immersion-based swimming.’ If it isn’t inquiry-based, it isn’t science.
Inquiry is essential in learning and doing science. The nature of science itself is an inquiry-based enterprise, and research has shown that students learn science best by doing science.
All national and state science standards include inquiry and/or the nature of science as prominent elements. The National Academy of Sciences has published a separate text titled “Inquiry and the National Science Education Standards (NSES): A Guide for Teaching and Learning,” which highlights the inquiry aspect of the NSES. In Rhode Island, where two Concord Consortium projects are taking place, the state standards, known as Grade Span Expectations, list inquiry and the nature of science as two of the six unifying themes across all science topics.
What is inquiry?
Inquiry has many different meanings. Some definitions include anything that involves a hands-on lab, while others are primarily focused on exploration of topics for which answers are unknown. Inquiry is possible at many levels, and it is this spectrum—from fully teacher centered to fully student centered— that is the source of much variability in defining inquiry. The following table provides a framework for categorizing an activity into a particular level of inquiry.
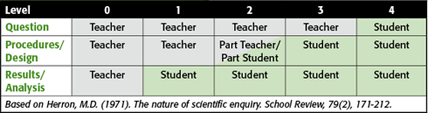
Level 0 describes an activity in which the teacher proposes the question, defines the methods and procedures, and analyzes the results for the students—for example, a classroom demonstration or a fill-in-the-blank lab experiment.
Level 4 describes an activity in which the student decides on the question to be explored, determines how to go about answering the question, and performs his or her own analysis of the data, including information from articles, discussions, and computer visualizations—as with a student project.
Depending on the practical limitations of time and equipment, as well as considerations regarding the best way to teach certain content, teachers will likely do activities across the inquiry spectrum.
Digital resources can support various levels of inquiry
A hands-on science lab could fall into any of these categories. But teachers can move even a Level 0 or 1 activity to a more student-centered approach by changing the language of the lab to be less prescribed and giving more control to the student in deciding how to conduct and analyze an experiment. Because digital materials are often not designed or easily modified by the teacher, it may seem less obvious how to use these tools at various levels of inquiry.
In the Rhode Island Information Technology Experiences for Students and Teachers (RI-ITEST) project, teachers do not have the ability to change the activities, which were developed to support an atomic-level understanding of physics, chemistry, and biology. However, teachers can adjust the way the activities are used for a more or less student-centered, inquiry-based approach.
The default mode for delivery of a RI-ITEST activity involves assigning students to complete an activity individually or in pairs. Students proceed through a sequence of pages containing models, instructions, challenges, and assessment items (Figure 1). The driving questions and procedures were created by the activity author. Students analyze the results and draw conclusions individually (or with a partner). Used in this way, an activity is a Level 1 or 2 on the inquiry scale depending on how structured or unstructured the model explorations are.
Class discussion and scientific reasoning
One aspect of the nature of science that is deeply entwined with inquiry is the ability to support an argument with evidence. The RI-ITEST teacher guides suggest models to highlight and discussion questions to facilitate full class conversation. For instance, try stopping the class as students are running the activity and have them support their claims by demonstrating how they came to a particular understanding using the models in the activity. This will reinforce their knowledge, build logical reasoning skills, enhance their ability to present ideas clearly—and move the activity up on the inquiry scale.
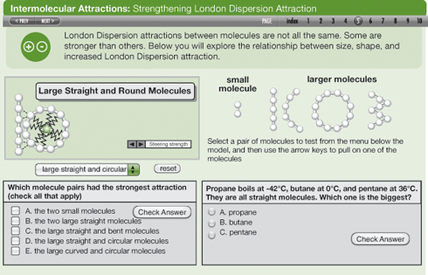
Alternatively, rather than having students do the activity individually, project the activity using an LCD projector. (If you don’t have access to enough computers for the entire class, this may be the best option in any case.) Show a model, then have students suggest ways to interact with it or invite one or more students to the projector or interactive whiteboard to engage with the model. Students can work together as a class to discover concepts and debate their interpretation of the resulting model behavior. Depending on how much the students contribute, this could be a Level 2 or 3 activity.
Customizing digital activities
Many of the digital resources developed at the Concord Consortium follow the pattern of a multi-page activity with embedded models, supporting text and images, and assessments. In some cases, as with our Information Technology in Science Instruction (ITSI) and Rhode Island Technology Enhanced Science (RITES) projects, teachers have the ability to edit existing activities or create new activities.

Using the Molecular Workbench (the software engine powering many of the molecular models) teachers and students can even design the models. This makes a Level 4 activity possible, where the student chooses a question to explore, designs models, and presents an analysis of the data. By designing her own model, a student was able to demonstrate how the number of molecules—not the number of atoms—is key to understanding volume of a gas (Figure 2).
Benefits of digital resources for inquiry
When I taught chemistry I told students that my most important lab rule was “no unauthorized experiments.” Rather than discouraging inquiry, I emphasized that I welcomed suggestions for experiments they wanted to try; they just had to get my approval first. I told them that 95% of the time I would approve any experiment they proposed, and the last 5% we would do together behind the safety shield.
Computer models allow students to have free range to explore extreme limits of the models without safety concerns. They can do many more experiments than in a wet lab, and the results are more immediate and easily shared with other students who can then reproduce the experiment. With the addition of digital resources to a curriculum, many opportunities for increased inquiry become possible.